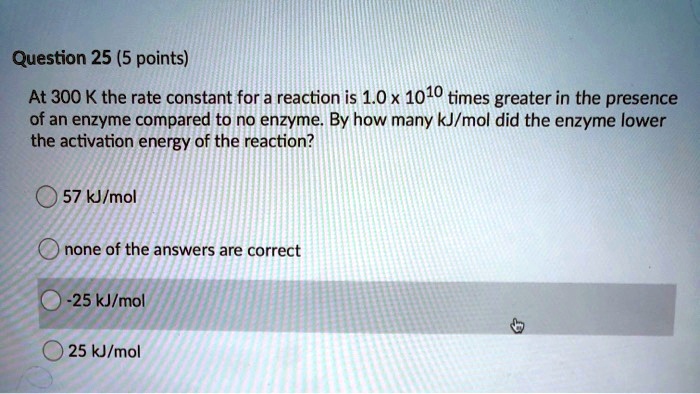
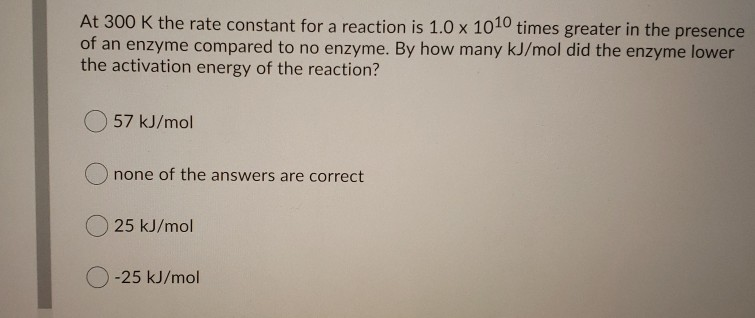SOLVED: Consider a general reaction: enzyme A(aq) + B(aq) The ΔGo of the reaction is -9.990 kJ mol^-1. Calculate the equilibrium constant for the reaction at 25 °C. Keq = 54.36 What

Enzyme Syrup In Prayagraj, Uttar Pradesh At Best Price | Enzyme Syrup Manufacturers, Suppliers In Allahabad

a) Activation energy ( Ea , kJ mol À 1 ), (b) Optimum temperature ( T... | Download Scientific Diagram

Enzymes involved in energy storage and energy supply by polyP. (A) ATP... | Download Scientific Diagram

The influence of pH on the rates of enzyme reactions. Part 5.—The case of several enzyme-substrate intermediates - Transactions of the Faraday Society (RSC Publishing)
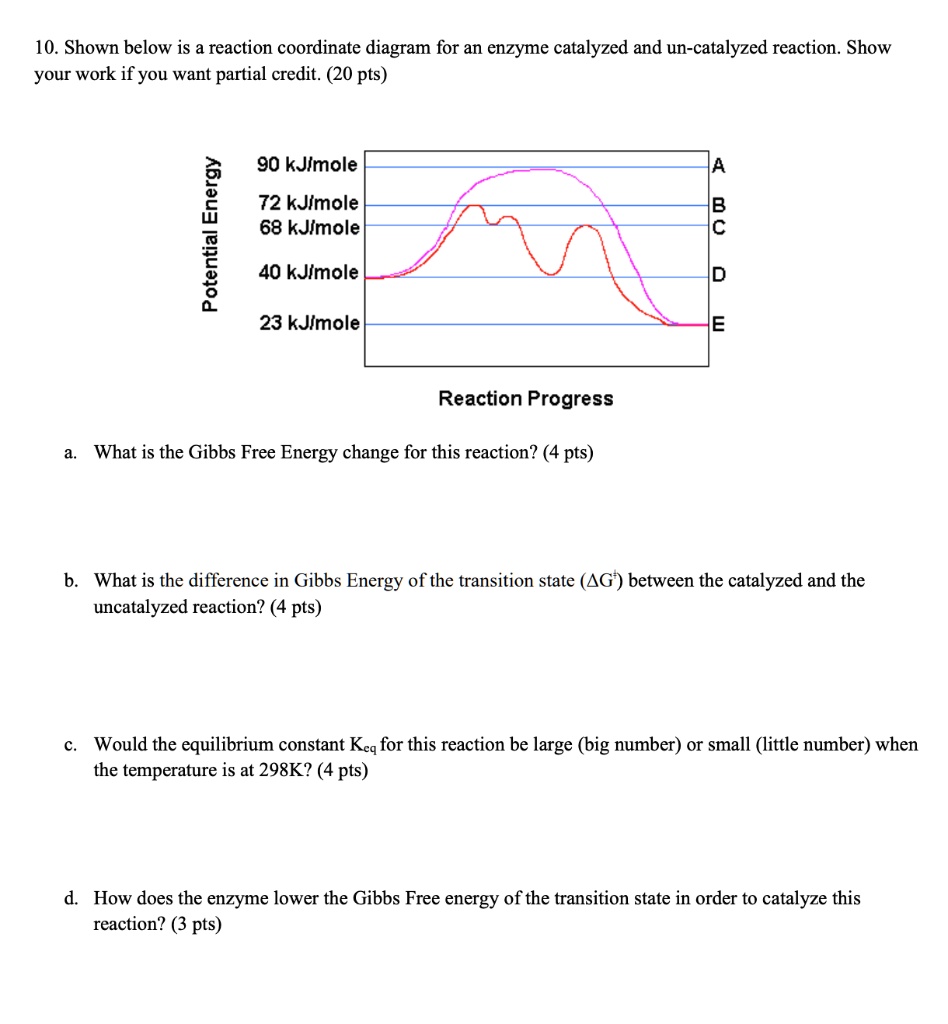
SOLVED: 10. Shown below is a reaction coordinate diagram for an enzyme-catalyzed and uncatalyzed reaction. Show your work if you want partial credit: (20 pts) 90 kJ/mol 1 72 kJ/mol 68 kJ/mol








![Values of activation energy, E a [kJ mol -1 ] and Q 10 value for... | Download Scientific Diagram Values of activation energy, E a [kJ mol -1 ] and Q 10 value for... | Download Scientific Diagram](https://www.researchgate.net/publication/334326868/figure/tbl3/AS:1049389002936321@1627205243258/Values-of-activation-energy-E-a-kJ-mol-1-and-Q-10-value-for-several-reactions.png)